
When you pick up a prescription, you might not realize that the pill in the bottle could be an authorized generic-a version of your brand-name drug that’s identical in every way, except for the label and packaging. Unlike regular generics, which are made by different companies and must prove they work the same way, authorized generics come from the exact same factory, with the same ingredients, same process, and same quality control as the original brand. The only difference? The name on the bottle.
What Makes an Authorized Generic Different
An authorized generic is not a copy. It’s the real thing, just sold under a different label. The brand-name company itself produces it, often through a subsidiary, and sells it to pharmacies at a lower price. This happens after the original patent expires but before other generic companies can enter the market. The FDA allows this under the Medicare Prescription Drug, Improvement, and Modernization Act of 2003. The first one appeared in 2004 when AstraZeneca released an authorized generic of Prilosec (omeprazole). Today, there are 147 authorized generics listed by the FDA as of October 2023.
Unlike traditional generics approved through the Abbreviated New Drug Application (ANDA) pathway, authorized generics are marketed under the original New Drug Application (NDA). That means they’re not just similar-they’re the same drug. Same active ingredient. Same inactive ingredients. Same manufacturing facility. Same quality standards. The only things that change are the label, the distributor name, and sometimes the color or imprint on the pill.
How to Spot an Authorized Generic by the Label
The label is your best clue. Look for these signs:
- No brand name-The drug won’t say "Lipitor" or "Lyrica." Instead, it’ll say the generic name like "atorvastatin" or "pregabalin."
- Distributed by-The manufacturer line will say "Distributed by [Company Name]" instead of "Manufactured by [Brand Name]." For example, if you see "Distributed by Greenstone LLC" on a pregabalin bottle, that’s an authorized generic of Lyrica, even though Pfizer made it.
- No trademark symbols-You won’t see ® or ™ next to the drug name. The label is stripped of marketing language and focuses only on the facts.
- Same pill appearance-Sometimes the pill looks identical. Other times, it’s a different color or shape. That’s because U.S. trademark laws prevent generics from looking exactly like the brand, even if they’re chemically identical.
A common mistake is assuming that if the pill looks the same, it’s the brand. But many authorized generics use identical pill designs. The real difference is in the packaging and labeling, not the medicine inside.
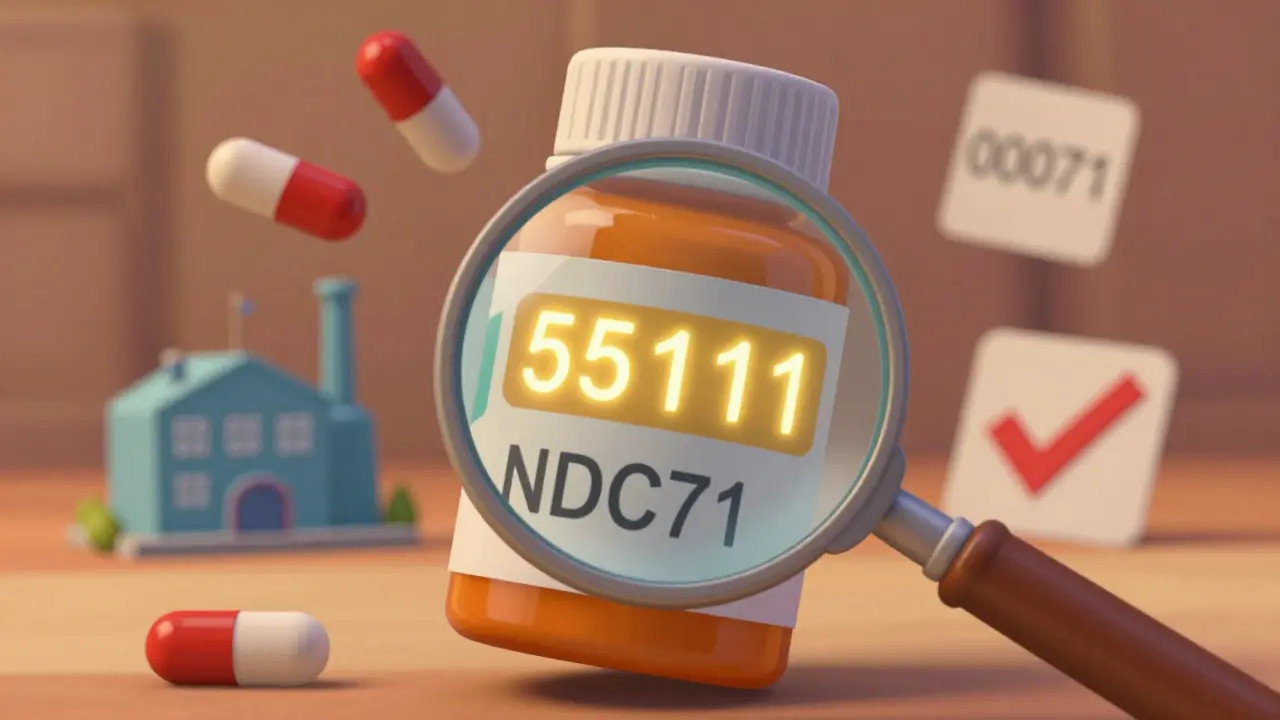
Check the NDC Number
The National Drug Code (NDC) is a unique 10- or 11-digit number on the packaging. It’s divided into three parts:
- Labeler code-The first set of numbers identifies the company that distributes the drug. For an authorized generic, this will be different from the brand name’s labeler code.
- Product code-The second set identifies the specific drug, strength, and dosage form. This will be identical to the brand.
- Package code-The third set tells you the package size (e.g., 30 pills, 90 pills). This also stays the same.
For example, the brand version of Protonix (pantoprazole) has an NDC starting with 00071. The authorized generic distributed by Dr. Reddy’s has an NDC starting with 55111-but the rest of the number is identical. If you compare the product and package codes and they match the brand, it’s an authorized generic.
The FDA updates its List of Authorized Generic Drugs quarterly. The most recent update was October 2, 2023. You can search this list to verify if the NDC you’re seeing is listed.
Why Authorized Generics Matter
Authorized generics offer a middle ground between brand-name drugs and regular generics. They’re usually priced 15-25% lower than the brand, but 5-15% higher than traditional generics. That’s because they’re not competing directly with other generics-they’re still sold under the brand’s distribution network.
For patients, the benefit is clear: identical effectiveness, lower cost, and no risk of bioequivalence uncertainty. A 2022 Medscape survey of over 1,200 patients found that 92.6% reported no difference in how the authorized generic worked compared to the brand.
For pharmacists, the challenge is identification. A 2022 survey by the National Community Pharmacists Association found that 63.4% of pharmacists took an average of 2.7 minutes per prescription to confirm if a drug was an authorized generic. That’s because packaging can be nearly identical to the brand, and many pharmacy systems don’t clearly flag them.
Common Mistakes and How to Avoid Them
Many people confuse authorized generics with other types of generics-or even counterfeit drugs. Here are the top errors:
- Thinking "same pill = same brand"-Just because the pill looks identical doesn’t mean it’s the brand. Check the label.
- Misreading "Manufactured by"-Authorized generics say "Distributed by." If it says "Manufactured by," it’s likely a traditional generic.
- Ignoring the NDC-Don’t rely on appearance. Always cross-check the NDC with the FDA’s official list.
- Confusing with "authorized brand"-Some companies sell traditional generics with brand-like packaging. These are not authorized generics. They’re just branded generics.
According to FDA data, 42.7% of labeling errors in generic submissions involve incorrect identification of authorized generic status. That’s why pharmacists need specialized training. The American Society of Health-System Pharmacists says it takes about 4.3 hours of focused learning to reliably identify them.

What to Do If You’re Unsure
If you’re confused about your prescription:
- Ask your pharmacist to check the NDC against the FDA’s Authorized Generic List.
- Look up the drug name and NDC on the FDA’s website or through your pharmacy’s database (First Databank or Medi-Span).
- Check the manufacturer name. If it’s a company you’ve never heard of, but the pill looks like your brand, it’s likely an authorized generic.
- Don’t assume it’s counterfeit just because the label looks different. Authorized generics are legal, safe, and regulated.
The FDA, FTC, and industry experts agree: authorized generics are a legitimate, high-quality option. They’re not a compromise. They’re the original drug, just without the brand name.
Are authorized generics as safe as brand-name drugs?
Yes. Authorized generics are made in the same facility, with the same ingredients, and under the same quality controls as the brand-name drug. The FDA inspects these facilities just like it does for brand-name manufacturers. There is no difference in safety or effectiveness.
Can I tell if my drug is an authorized generic just by looking at the pill?
Not always. Sometimes the pill looks exactly the same. Other times, it’s a different color or shape due to trademark rules. The only reliable way is to check the label for "Distributed by" and compare the NDC number to the FDA’s Authorized Generic List.
Why do authorized generics cost more than regular generics?
Because they’re sold by the brand-name company’s own distribution network, often with less competition. Regular generics compete with dozens of other manufacturers, driving prices down. Authorized generics have less competition, so they’re priced higher than traditional generics but still lower than the brand.
Do authorized generics show up in the FDA’s Orange Book?
No. The Orange Book only lists drugs approved through the ANDA pathway. Authorized generics are marketed under the original NDA, so they don’t appear there. Instead, they’re listed separately on the FDA’s Quarterly Authorized Generic List.
Is it true that some authorized generics have "AG" on the label?
Some do, but it’s not required. The FDA doesn’t mandate any special markings. A few companies add "Authorized Generic" or "AG" in small print as a courtesy, but many don’t. Don’t rely on this-it’s not a rule.
Next Steps for Patients and Pharmacists
If you’re a patient: Always ask your pharmacist if your prescription is an authorized generic. If it is, you’re getting the exact same drug at a lower price. No need to worry.
If you’re a pharmacist: Use the FDA’s Authorized Generic List as your primary reference. Train your team to check the NDC labeler code, not just the pill appearance. Update your pharmacy system to flag authorized generics clearly.
By 2024, the FDA plans to integrate authorized generic identifiers directly into the National Drug Code Directory. That will make identification easier. Until then, the best tool you have is knowledge-and a quick look at the label and NDC number.










Just learned about authorized generics last week and it blew my mind. I’ve been taking omeprazole for years and never realized the bottle changed but the pill inside was the exact same. No wonder my prescription cost dropped by 40%.
Pharmacists should be required to explain this at pickup. It’s not just about saving money-it’s about knowing you’re getting the real thing, not some sketchy copy.
Ugh. So... let me get this straight. You're telling me the FDA lets big pharma sell their own drug under a different label... and call it a 'generic'? That's not a generic. That's a loophole. And don't even get me started on how they market it as 'better' than real generics. Please. The whole system is rigged.
Authorized generics are actually a smart move. Brand companies make more sense to produce them-same facility, same QC, same batch. No need for ANDA approval. Saves time, saves cost. Real generics? Sometimes you get variability. Not here. The NDC check is key. I always cross-check with the FDA list now. Works every time.
This is the kind of info that changes lives. If you're paying full price for a brand when you could be getting the same medicine for less, you're leaving money on the table. Simple as that. Ask your pharmacist. Know your NDC. Take control.
Just checked my last script-Distributed by Greenstone. Same pill as the brand. Saved $38 this month. This is the kind of thing we should all be talking about more.
Let me guess. Big Pharma is behind this. They know people will trust 'same factory' and think it's safer. But here's the truth: they're using this to delay real competition. They create an authorized generic, lock in the market, then sit back while cheaper generics are held up by red tape. This isn't transparency-it's manipulation disguised as savings.
And don't tell me about the FDA list. I've seen NDCs get updated three months after the fact. You think your pharmacist is checking that? Ha. They're scanning barcodes and hoping.
I love how this post breaks it down. I used to panic when my pill changed color. Now I just look for 'Distributed by' and check the NDC. It’s so simple once you know what to look for.
My mom’s on 12 meds. I helped her switch three to authorized generics. Her out-of-pocket dropped by $200/month. She’s not a pharmacist. She just needed someone to show her how.
Anyone who thinks this is 'the real thing' is naive. The brand company still owns the patent. They still control the supply. They still profit off the same product with a different label. This isn’t competition. It’s a controlled release to soften the blow of generic entry. They’re not lowering prices to help you-they’re lowering them just enough to keep you from switching to a real competitor.
Imagine if your favorite coffee shop started selling the exact same beans in a plain bag with no logo-same roast, same grind, half the price. You’d be thrilled. That’s what authorized generics are. No fluff. No marketing. Just the good stuff, stripped down to what matters.
Pharmacists are the unsung heroes here. They’re the ones who catch it, explain it, and make sure you don’t get screwed. We need more of them.
I come from South Africa where generics are a lifeline. We don’t have the luxury of brand-name drugs. So when I heard about authorized generics in the US, I was skeptical. But the data? Solid. Same factory. Same standards. If it works here, it should work everywhere. This is a model worth exporting.
I cried when I found out my anxiety med was an authorized generic. I thought I was being scammed. Turns out I was being saved. Thank you for explaining this so clearly. I’m telling everyone.
My pharmacist just told me my new bottle of amoxicillin was an authorized generic. I thought she was messing with me. Then I checked the NDC. Same product code as the brand. Just distributed by a different company. I didn’t even notice the difference. That’s the whole point, isn’t it?